Atom and Molecules Full Chapter Notes
(Easy + Detailed Guide)
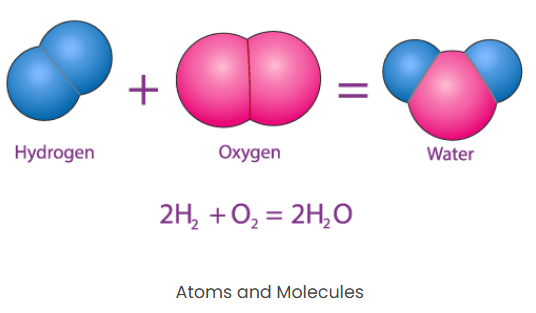
Atoms and Molecules
Question: What is an atom?

Answer: An atom is the smallest particle of any element. An important property of an atom is that it retains the chemical properties of that element.
Question: What is a molecule?
Answer: When two or more atoms combine with one another, they form a molecule.
Question: Who proposed the Law of Conservation of Mass?
Answer: The Law of Conservation of Mass was proposed by Antoine Lavoisier.
Question: What is the Law of Constant Proportions?
Answer: In any given compound, elements are always present in a fixed proportion by mass.
Question: What is a chemical formula?
Answer: The symbolic representation used to depict a chemical compound is called a chemical formula.
Question: Define the term “mole.”
Answer: A mole is defined as the amount of a substance that contains 6.022 × 10²³ particles.
Question: Define 1 amu.
Answer: 1 amu is defined as exactly one-twelfth (1/12) of the mass of a single carbon-12 atom.
Question: Can atoms be seen directly?
Answer: No, we cannot see atoms directly.
Question: Does the total mass change during a chemical reaction?
Answer: The total mass never changes during a chemical reaction.
Question: Explain the Law of Conservation of Mass.
Answer: Law of Conservation of Mass—According to this law, in any chemical reaction, the total mass can neither be created nor destroyed; however, it can be transformed from one form into another. This is known as the Law of Conservation of Mass.
Question: Explain the Law of Constant Proportions, and provide an example as well.
Answer: According to this law, in any chemical compound, elements are always present in a fixed proportion.
Example: In water, the ratio of hydrogen to oxygen remains 2:1.Atom and Molecules
Full Chapter Notes (Easy + Detailed Guide)
Question: Define an ion.
Answer: When an atom gains or loses electrons, it becomes an ion.
[Long Answer]
Question: Explain the Law of Conservation of Mass.
Answer: Law of Conservation of Mass—According to this law, in any chemical reaction, the total mass can neither be created nor destroyed; however, it can be transformed from one form into another. This is known as the Law of Conservation of Mass.
Question: Explain the Law of Constant Proportions, and provide an example as well.
Answer: According to this law, in any chemical compound, elements are always present in a fixed proportion.
Example: In water, the ratio of hydrogen to oxygen remains 2:1.
Question: Define an ion.
Answer: When an atom gains or loses electrons, it becomes an ion.
Question: Explain Dalton’s Atomic Theory in detail.
Answer: Dalton’s Atomic Theory—John Dalton presented a significant theory regarding atoms, which laid the foundation for modern chemistry.
Its main points are as follows:
-
All matter is composed of atoms.
-
Atoms are indivisible.
-
Atoms of the same element are identical.
-
Atoms of different elements are distinct.
-
In a chemical reaction, atoms are merely rearranged.
Question: Define the ‘mole’.Answer: The mole is a unit of measurement used to represent the number of microscopic particles.
Example: One mole is equal to 6.022 × 10²³.
Question: Explain the difference between atomic mass and molecular mass.
Answer: Atomic mass is the mass of a single atom, whereas molecular mass is the sum of the masses of all the atoms constituting an entire molecule.
Question: Explain atoms and molecules in detail.
Answer: Atoms and molecules are among the most important and fundamental concepts in chemistry. Knowledge of these two concepts is essential for understanding the structure and behavior of any substance. If we observe the world around us, we see various objects existing in solid, liquid, or gaseous states; all of these are composed of microscopic particles. These microscopic particles are precisely what are referred to as atoms and molecules. In ancient times, Indian and Greek philosophers proposed the idea that matter is composed of tiny, indivisible particles.

The Indian philosopher Kanada stated that the atom is an extremely small particle, while the Greek philosopher Democritus named these particles ‘atomos’. Although this was merely a philosophical concept at the time, it was later scientifically proven through experiments conducted by scientists. An atom is the smallest unit of any element that retains the chemical properties of that element. If we repeatedly divide an element, we eventually arrive at an atom. However,
if we were to divide the atom itself, it would lose its fundamental properties. Therefore, the atom is considered the fundamental unit of matter. The atoms of every element are distinct from one another. For example, the hydrogen atom is the lightest atom, whereas the uranium atom is very heavy. This is precisely why different elements possess different properties; the mass, size, and structure of an atom determine its characteristics. Question: Explain Dalton’s Atomic Theory.
Answer: Dalton’s full name was John Dalton; he was a renowned scientist who was the first to present the theory of the atom on a scientific basis. His atomic theory is considered the backbone of modern chemistry. He endeavored to explain how matter is composed of tiny particles and how these particles participate in chemical reactions.
According to Dalton, all matter is composed of extremely small particles known as atoms. These atoms are so minute that they cannot be perceived by the naked eye. He further stated that atoms are indivisible—meaning they cannot be broken down into smaller parts. Although scientists subsequently demonstrated that sub-atomic particles exist within the atom itself, Dalton’s theory was of immense significance at the time it was proposed.
Dalton also asserted that all atoms of a specific element are identical; they possess the same mass, size, and properties. Conversely, atoms belonging to different elements are distinct from one another. This distinction accounts for the fact that the properties of each element are unique.
According to Dalton’s theory, atoms combine in simple whole-number ratios to form compounds. For instance, in water, the ratio of hydrogen to oxygen is fixed. This theory proved to be highly instrumental in facilitating an understanding of chemical reactions.
However, Dalton’s theory did possess certain limitations. He posited that the atom was indivisible—a premise that was later proven incorrect. Furthermore, he did not provide an explanation regarding the internal structure of the atom. Nevertheless, despite these limitations, his theory remained profoundly significant.
Dalton’s theory proved to be a milestone in the evolution of chemistry. It inspired scientists to pursue further research and laid the foundation for the modern theory of atomic structure.
Important Topic Read>
1.Matter in Our Surroundings Notes
