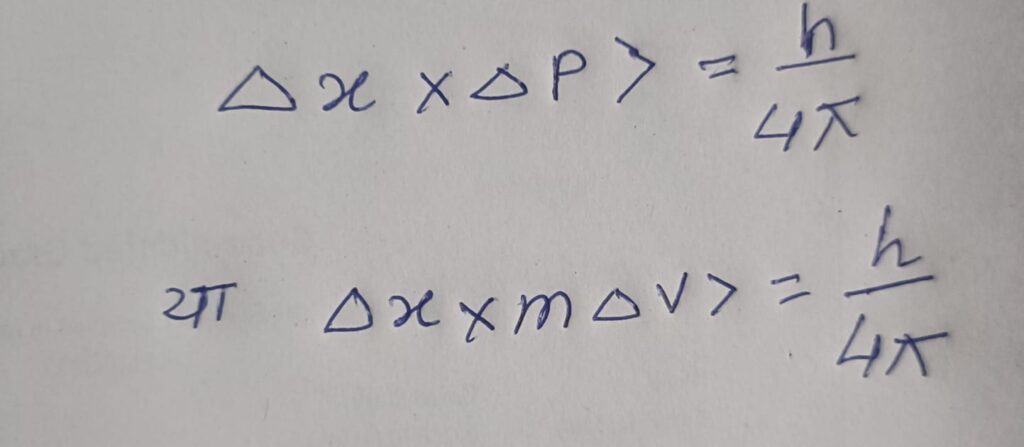Class 12 Nios Board Chemistry Chapter 2nd-Atomic Structure
Explain the atomic structure.
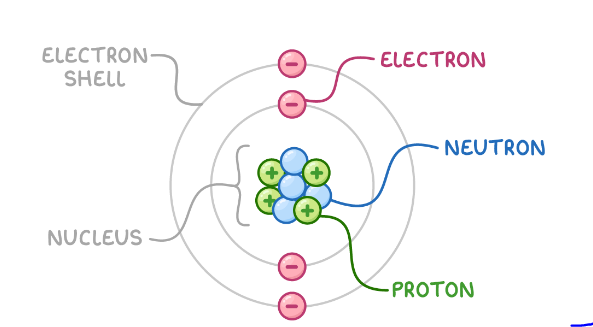
Answer – The atomic structure of an element describes the number of protons and electrons in its nucleus and the number of electrons surrounding it. Essentially, the atomic structure of matter is composed of protons, electrons, and neutrons.Class 12 Nios Board Chemistry Chapter 2nd-Atomic Structure Protons and neutrons form the nucleus of the atom. The atomic number of an element indicates the total number of protons in its nucleus.
Atomic Models:
In the 18th and 19th centuries, many scientists attempted to understand the structure of the atom with the help of atomic models. Each of these models had its own advantages and disadvantages and was crucial for the development of the modern atomic model. Scientists like John Dalton, J.J. Thomson, Ernest Rutherford, and Niels Bohr contributed to this development.
Explain Dalton’s Atomic Theory.
Answer – Dalton was an English chemist. Dalton suggested that all matter is made up of atoms, which are indivisible and indestructible. He also stated that all atoms of an element are exactly alike, but atoms of different elements differ in size and mass.
According to Dalton’s atomic theory, chemical reactions involve the rearrangement of atoms to form products. Class 12 Nios Board Chemistry Chapter 2nd-Atomic Structureb According to the postulates proposed by Dalton, an atom can participate in any chemical reaction.
Important Postulates of Dalton’s Theory:
1. Every substance is made up of atoms. Atoms are indivisible.
2. Specific elements contain only one type of atom.
3. Each atom has its own stable mass, which differs from one element to another.
4. Atoms undergo rearrangement during chemical reactions.
Atoms can neither be created nor destroyed, but can be transformed from one form to another. Dalton’s Atomic Theory Defects:
1. Dalton was unable to explain the existence of isotopes.
2. Dalton could not accurately describe anything about the structure of the atom.
3. Scientists discovered particles inside the atom that proved that the atom is divisible.
1. Thomson’s Atomic Model
The English chemist Sir Joseph John Thomson presented his model describing the atomic structure in the early 1900s. He was later awarded the Nobel Prize for the discovery of the electron.
NOTE – His work is based on an experiment called the cathode ray experiment.
Question – Explain Rutherford’s atomic theory.
Answer – Rutherford modified the atomic structure with the discovery of another subatomic particle called the nucleus, based on an experiment by J.J. Thomson. His atomic model was based on the alpha ray scattering experiment.
Alpha Ray Scattering Experiment –
Construction: He took a very thin gold foil, 1000 atoms thick. Alpha rays (doubly charged He²⁺) were made to bombard the gold foil.
A ZnS screen was placed behind the gold foil. Most of the rays passed through the foil, creating bright spots, while some rays collided with the foil and were then deflected.
One in 1000 rays collided with the gold foil and came back at an angle of 180 degrees.
4. Explain Rutherford’s atomic structure –
Answer – Rutherford proposed his own atomic structure, which is as follows:
1. The nucleus is at the center of an atom, where most of the charge and mass are located.
2. The atomic structure is spherical.
3. Electrons revolve around the nucleus in a circular orbit, similar to how planets orbit the sun.
Explain Bohr’s atomic theory.
Answer: Niels Bohr put forward his model of the atom in 1915. Even today, it is used to provide a comprehensive explanation of the element. This atomic model is based on Planck’s quantization principle. Electrons within atoms are placed in discrete orbits, called stationary orbits.
1. The energy levels of these shells are represented by quantum numbers.
2. As long as an electron remains in its stationary orbit, it does not absorb or emit energy.
3. Electrons revolve around the nucleus only in these stationary orbits.
4. The energy of the stationary orbits is quantized.
Limitations of Bohr’s atomic theory:
Limitations of Bohr’s Atomic Theory
1. Bohr’s atomic model works only for single-electron species.
EXAMPLE – H, He⁺, Li²⁺, Be³⁺
2. When the emission spectrum of H₂ was observed under a more precise spectrometer,
3. Neither the Stark effect nor the Zeeman effect can be explained using Bohr’s theory.
Explain Heisenberg’s Uncertainty Principle?
Answer – The electron exhibits both particle and wave nature. And the exact position of a wave cannot be determined.
1. If the exact position of the wave is determined with 100% accuracy, there will be a difference in the value of the wave and the particle.
2. This principle was given by the German physicist Werner Heisenberg. Therefore, “It is impossible to accurately determine the position and momentum of a subatomic particle simultaneously.” Mathematically-